 |
|
|
|
|
|
|
| ||||||||||
|
|
|
|
|
|
|
||||
| ||||||||||
|
|
|
|
|
Persons using assistive technology might not be able to fully access information in this file. For assistance, please send e-mail to: [email protected]. Type 508 Accommodation and the title of the report in the subject line of e-mail. Interpreting and Managing Blood Lead Levels <10 µg/dL in Children and Reducing Childhood Exposures to LeadRecommendations of CDC's Advisory Committee on Childhood Lead Poisoning PreventionPlease note: An erratum has been published for this article. To view the erratum, please click here. Prepared by
The material in this report originated in the National Center for Environmental Health/Agency for Toxic Substances and Disease Registry, Howard Frumkin, MD, DrPH, Director; and the Division of Environmental and Emergency Health Services, Sharrunda Buchanan, PhD, Director. Corresponding preparer: Mary Jean Brown, ScD, Division of Environmental and Emergency Health Services, National Center for Environmental Health/Agency for Toxic Substances and Disease Registry, CDC. Telephone: 770-488-7492; Fax: 770-488-3635; E-mail: [email protected]. AbstractLead is a common environmental contaminant, and exposure to lead is a preventable risk that exists in all areas of the United States. Lead is associated with negative outcomes in children, including impaired cognitive, motor, behavioral, and physical abilities. In 1991, CDC defined the blood lead level (BLL) that should prompt public health actions as 10 µg/dL. Concurrently, CDC also recognized that a BLL of 10 µg/dL did not define a threshold for the harmful effects of lead. Research conducted since 1991 has strengthened the evidence that children's physical and mental development can be affected at BLLs <10 µg/dL. This report summarizes the findings of a review of clinical interpretation and management of BLLs <10 µg/dL conducted by CDC's Advisory Committee on Childhood Lead Poisoning Prevention. This report provides information to help clinicians understand BLLs <10 µg/dL, identifies gaps in knowledge concerning lead levels in this range, and outlines strategies to reduce childhood exposures to lead. In addition, this report summarizes scientific data relevant to counseling, blood lead screening, and lead exposure risk assessment. To aid in the interpretation of BLLs, clinicians should understand the laboratory error range for blood lead values and, if possible, select a laboratory that achieves routine performance within ±2 µg/dL. Clinicians should obtain an environmental history on all children they examine, provide families with lead prevention counseling, and follow blood lead screening recommendations established for their areas. As local and patient circumstances permit, clinicians should consider early referral to developmental programs for children at high risk for exposure to lead and consider more frequent rescreening of children with BLLs approaching 10 µg/dL, depending on the potential for exposure to lead, child age, and season of testing. In addition, clinicians should direct parents to agencies and sources of information that will help them establish a lead-safe environment for their children. For these preventive strategies to succeed, partnerships between health-care providers, families, and local public health and housing programs should be strengthened. IntroductionLead is a common environmental contaminant, and exposure to lead is a preventable risk in all areas of the United States. Lead is associated with negative outcomes in children, including impaired cognitive, motor, behavioral, and physical abilities (1--4). In 1991, CDC defined the blood lead level (BLL) that should prompt public health actions as 10 µg/dL. Concurrently, CDC also recognized that a BLL of 10 µg/dL did not define a threshold for the harmful effects of lead (5). Research conducted since 1991 has strengthened the evidence that children's physical and mental development can be affected at BLLs <10 µg/dL (1,3). During 2002--2004, a workgroup of CDC's Advisory Committee on Childhood Lead Poisoning Prevention (ACCLPP) reviewed the scientific literature regarding adverse health effects associated with BLLs <10 µg/dL, including 23 published reports that analyzed 16 separate populations with Intelligence Quotient (IQ) or general cognitive index outcomes and 12 publications related to other health outcomes. In its 2005 report, the workgroup concluded that an inverse association exists between BLLs and cognitive function, with no evidence of a weaker association in populations with lower BLLs (1). The direct evidence for this inverse association was strongest in a study conducted in Rochester, New York, that included children born in 1994 and1995, enrolled at age 6 months, and followed for 5 years (6). The majority of children studied had BLLs <10 µg/dL throughout the study period. The IQ and blood lead level relationship was most accurately described by a nonlinear negative association, with a decrease in IQ of more than seven points over the first 10 µg/dL increase in lifetime average blood lead concentration. On the basis of the evidence, the workgroup concluded that a causal association between lead exposure and impaired cognitive functioning was most likely. However, the potential for residual confounding, particularly by social factors, made the strength and shape (i.e., linear or nonlinear) of this association across BLLs uncertain. In addition, the workgroup concluded that children with BLLs <10 µg/dL should not be classified as "lead poisoned." The report noted that no safe level for blood lead in children has been identified (1). Two studies published subsequently have reported negative effects of BLLs <10 µg/dL on developmental outcomes (7,8). One study, which included participants from the Rochester cohort (6) and from six other past prospective studies of children with peak BLLs across a range of values, reaffirmed an inverse association between lead at low levels and IQ (7). In these studies, key potential confounders were accounted for, including maternal IQ, the Home Observation for Measurement of the Environment Inventory (HOMEI) score (which is a measure of the quality and quantity of stimulation and support available to a child in the home environment), maternal education, and birth weight. Although ACCLPP has previously reviewed case management of children with BLLs >10 µg/dL (2), this is the first ACCLPP report to summarize scientific information relevant to clinical management of children with BLLs <10 µg/dL. This report also outlines recommendations from ACCLPP to reduce childhood exposure to lead. Information on assessing an environmental history and prevention strategies to decrease exposures to lead have been published previously (2,3) and are not included in this report. MethodsACCLPP provides advice and guidance to the U.S. Department of Health and Human Services and CDC regarding new scientific knowledge and technologic developments and their practical implications for preventing childhood lead poisoning, and recommends improvements, as needed. ACCLPP members are selected on the basis of their expertise in childhood lead poisoning prevention, blood lead screening, diagnosis, and medical management. ACCLPP liaisons represent federal agencies and organizations with particular interest and expertise in childhood lead poisoning prevention. In October 2003, ACCLPP formed another workgroup comprising three pediatricians and a CDC health scientist to review the scientific literature regarding clinical management options for BLLs <10 µg/dL and to outline recommendations for clinical care providers. On the basis of its analysis, the workgroup developed draft recommendations that were reviewed and later adopted by ACCLPP in February 2006. ResultsHistoric Trends in Children's BLLs in the United StatesSince 1976, BLLs in U.S. children aged 1--5 years have decreased substantially (Table 1), primarily as a result of policies that have reduced the dispersal of lead into the environment (9--12). However, many U.S. children continue to be exposed to lead, primarily in their homes (13). Overt clinical symptoms of lead intoxication are uncommon in the United States, and lead evaluation and management strategies typically are intended to reduce the negative effects of lead on central nervous system development in children who are clinically asymptomatic. Because no safe BLL has been defined (1), small reductions in population-level exposures to lead will likely affect substantial numbers of children, and can be expected to reduce the number of children affected by adverse health outcomes associated with lead exposure (14). Blood Lead MeasurementsAs with any biologic test, blood lead measurements entail inherent uncertainties as a result of imprecise analytic techniques and preanalytic variables (e.g., the specimen collection process). However, the ratio of imprecision to measurement value, particularly at BLLs <10 µg/dL, is relatively high. The degree of inherent error in blood lead analysis varies by analytic methodology used, but whichever method is used, laboratory performance depends on the procedures and skills of the laboratory team (15,16). Federal regulations allow laboratories that perform blood lead testing to operate with a total allowable error of ±4 µg/dL or ±10%, whichever is greater. Consequently, at BLLs <10 µg/dL, a laboratory might operate within an error range of 8 µg/dL and still meet federal proficiency standards. For example, an actual value of blood lead at 7 µg/dL could be reported as being any value ranging from 3 µg/dL to 11 µg/dL and still remain within the allowable error limit. A study of duplicate testing of identical blood samples (all with a mean blood lead value <10 µg/dL) at eight laboratories reported all results as <10 µg/dL and within 3 µg/dL of the overall mean for that specimen value (17). A study conducted in 2006 indicated that the majority of blood lead laboratories can achieve routine performance of ±2 µg/dL at concentrations of <10 µg/dL without difficulty (18). Blood lead test reliability also depends on adhering to blood collection techniques that reduce sample contamination. Collection of capillary blood from a fingerstick into a lead-free collection device is an accepted method for obtaining a screening test (19--23) and contamination by lead from the skin surface can be minimized if a protocol for proper capillary specimen collection is followed (24).† However, because lead levels from a capillary blood sample will vary from those of a simultaneously drawn venous sample, elevated capillary results should be confirmed with blood drawn by venipuncture. Multiple studies have reported on the uncertainty introduced by collecting capillary blood rather than venipuncture at thresholds of 10 µg/dL or 15 µg/dL (19--23), but none has examined the sensitivity or specificity of capillary methods at thresholds <10 µg/dL. Children's BLL PatternsBLLs increase quickly after an acute exposure, then gradually (over weeks) reach equilibrium with body stores of lead. Lead is distributed unevenly within the human body; in children, approximately 70% is stored in the bone compartment (25--27). The residence time of lead in bone can be decades (28). Thus, an elevated BLL will decline within a few weeks to months after an acute exposure. However, for those children with chronic lead exposure and, presumably higher bone lead stores, the decline in BLL can take much longer (29). Although bone lead levels can provide information regarding past absorption of lead, measurements of lead in bone using X-ray fluorescence instruments are available for research purposes only. A newborn infant's BLL closely reflects that of the mother (30). During 1999--2002, the geometric mean BLL for U.S. women aged 20--59 years was 1.2 µg/dL, with 0.3% having a BLL >10 µg/dL (12). Typically, as infants become more active and increase their environmental exposures, BLLs increase. Longitudinal studies of lead-exposed children have confirmed an increase in BLLs beginning in late infancy, with a peak level reached at age 18--36 months (6,31--33). No studies have examined blood lead patterns specifically for children with peak levels <10 µg/dL, although certain studies have included children with levels this low. A study of children born during 1994--1995 in which >50% of the children had peak BLLs <10 µg/dL reported an expected pattern in mean BLLs of 3.4 µg/dL at age 6 months, 9.7 µg/dL at age 24 months, and 5.8 µg/dL at age 61 months (6). A study of children born in Boston during 1979--1981 identified mean BLLs of 7.2 µg/dL at birth, and subsequent BLLs in these children remained relatively constant (6.2 µg/dL at age 6 months, 6.8 µg/dL at age 24 months, and 6.4 µg/dL at age 57 months) (34--36). In both studies, higher levels of lead in home environmental samples were associated directly with higher BLLs in children (35,37). In addition, the Boston study demonstrated an association between the occurrence of home renovation and increased BLLs (35). The blood lead pattern for individual children with BLLs <10 µg/dL varies depending on their environmental exposures (29). More research is needed to better understand age-related patterns for BLLs that remain <10 µg/dL. However, in clinical practice, even should additional research data become available, laboratory uncertainty might interfere with a clinician's ability to detect patterns for individual children. Once a high BLL has been established in a child, the time required for the BLL to decline to <10 µg/dL can range from months to years, depending on the duration and dose of exposure. For example, for a group of children starting at a BLL of 10--14 µg/dL and receiving case management services, the mean time required for 50% to achieve a BLL <10 µg/dL was 9 months (38). How much time is needed for BLLs <10 µg/dL to decline in response to interventions is unknown. Multiple studies have confirmed that blood lead measurements vary seasonally. For example, a study conducted in Boston reported that BLLs were highest in late June and lowest in March (39). A Milwaukee study indicated that BLLs were higher in the summer than in the winter (40). Some of the variability (higher blood lead in summer) might result from increased exposure to lead in dust and soil in summer months (41). Blood lead values for urban children are predicted to be 1--2 mg/dL higher in the summer than winter months (42). Association of BLL Patterns with Developmental OutcomesAlthough BLLs peak in early childhood, when young children are especially vulnerable to lead, negative effects are associated with lead exposure at any age. Multiple studies have examined the effects of lead on children's development outcomes; in these studies, the ages at which BLLs were measured varied, as did the range of ages over which BLLs were averaged (1--4). Statistically significant associations have been identified between average BLLs over a specific period (e.g., 0--5 years) and various adverse health outcomes (6,43--45); other studies have reported statistically significant associations with a single lead measurement at a specific age (e.g., prenatal, 24 months, and 6.5 years) or with a peak measurement (6,31,46). Concurrent BLLs (i.e., those measured close to the time of neurodevelopmental testing) might demonstrate stronger associations with neurodevelopmental abilities than other blood lead measures (6--8,32,47). Lead has a continuing negative association with IQ as children reach elementary school age. For children who participated in a trial of chelation therapy, a subsequent data analysis indicated that BLLs measured concurrently with developmental testing were associated more closely with children's cognitive abilities than was a peak level at approximately age 2 years (48). This association was stronger when children were tested at age 7 years than at age 5 years, which underscores the continuing need to reduce lead exposures after age 5 years. Strategies to Enhance Children's Positive Developmental OutcomesAlthough lead is a risk factor for developmental and behavior problems, its presence does not indicate that these problems will necessarily occur. No characteristic developmental pattern is attributable solely to the effects of lead, and measures of the effects of lead on children are imperfect. Thus, for an individual child, neurobehavioral test performance might indicate clinically-significant impairments related to lead exposures but might not fully capture the array of negative outcomes caused by lead (14). The effects of lead at levels approaching 10 µg/dL might not be recognizable to either the child's family or clinician or be identified through neurobehavioral testing. However, lead exposure might assume greater importance for children with other environmental, genetic, biologic, social, or demographic developmental risks factors. Effects of exposures to lead at lower levels might not be evident in testing of individual children but are best evaluated on a communitywide basis (14). Multiple factors influence a child's development, including how the child is treated by parents or other adult caregivers. The child's family and personal psychosocial experiences are strongly associated with performance on neurodevelopment measures and account for a greater proportion of the explained variance in these measures than BLLs <10 mg/dL(2,43,45,49). A child's blood lead measurement is estimated to account for 2%--4% of variance in neurodevelopment measures (approximately 4%--8% of the explained variance) (2,43,50). All children benefit from parental nurturing, regardless of their BLL. For example, a child's language skills are enhanced by the amount of language addressed to the child (more is better), combined with a predominant pattern of positive feedback (51). This pattern of parenting of children under age 3 years has been associated with enhanced language and cognitive skills when children were tested in the third grade (52). Thus, parents might help counteract the negative effects of lead by providing a nurturing and enriched environment during development. Studies to examine effects of lead have attempted to control for this psychosocial factor by including measures such as the HOMEI score (7). Although no studies have specifically evaluated the effects of early intervention programs on cognitive or behavioral outcomes in relationship to children's BLLs, several laboratory studies that applied a nurturing environment to very young animals during lead acquisition demonstrated the beneficial effect of the social environment on ameliorating lead-related negative developmental outcomes (53,54). Early enrichment programs, although not tested specifically in relation to BLLs, have been effective in improving cognitive development and social competence of young children, particularly infants from families with low levels of social or economic resources (55). Research demonstrates that children whose development has been delayed or who are at high risk for delay benefit most from interventions applied at an early age (56--58). Strategies to Prevent and Reduce Exposure to LeadCDC and the American Academy of Pediatrics (AAP) recommend that preventive care for every child should include obtaining an environmental history and identifying occupational lead exposure of household members (2,3,5). The major sources of lead exposure among U.S. children are lead-contaminated dust, deteriorated lead-based paint, and lead-contaminated soil (37,59). Typically, lead contamination of water contributes less to a child's lead burden than home and soil sources (59); however, if additives to water (e.g., those used in disinfection processes), are changed, the contribution of lead contamination might be greater (60). The extent of lead paint hazards (i.e., the presence of lead in an accessible condition, such as deteriorated lead-based paint or lead-contaminated dust or soil) on interior and exterior surfaces and in soil is associated with increased BLLs in children (59). Children also are exposed to nonhousing lead sources (e.g., lead in foods, cosmetics, pottery, folk remedies, and toys) (2,3,61). Home-Related Lead Exposure An estimated 4.1 million homes in the United States (25% of U.S. homes with children aged <6 years) have a lead-based paint hazard (13). An estimated 68% of U.S. homes built before 1940 have lead hazards, as do 43% of those built during 1940--1959 and 8% of those built during 1960--1977; estimates are higher for homes in the Northeast and Midwest and for homes in which young children reside (13). Despite considerable attention and resources from federal, state, and local agencies and advocacy groups, publicly available funding has not been able to provide sufficient resources to eliminate all lead paint hazards from U.S. homes. Publicly funded home inspections are most often limited to homes of children with elevated BLLs; the blood lead threshold value that prompts an inspection varies by state or municipality (62). In addition, even when a child's elevated BLL triggers an inspection, public funding for repairs to reduce or eliminate identified lead hazards typically is not available. Since 1991, lead-hazard--control grant programs through the U.S. Department of Housing and Urban Development's (HUD) Office of Healthy Homes and Lead Hazard Control (OHHLHC) have provided funding for local and state agencies to reduce lead and other environmental hazards in privately owned low-income housing. In 2005, OHHLHC allocated $139 million for this purpose, administered through seven different grant types. Other federal programs provide funding to eliminate lead-based paint hazards in federally assisted housing. Typically, the focus of these programs is on housing rehabilitation and activities that remediate lead hazards after children are identified with elevated BLLs, but HUD-funded local programs also now include primary prevention interventions that control or eliminate lead before children are exposed. CDC is working with HUD, the U.S. Environmental Protection Agency (EPA), state and local health department lead poisoning prevention grantees, and child health and environmental justice advocates to promote primary prevention strategies to reduce exposure to lead (1,63,64). In addition to their traditional role of providing services to children with elevated BLLs, CDC-funded state and local lead poisoning prevention programs have been charged with implementation of housing-based primary prevention strategies in their jurisdictions. This moves beyond their traditional role of providing services to children with elevated BLLs and involves developing responses to local risks and a focus on identifying and remediating housing-based lead hazards. ACCLPP recommendations for essential elements for state and local primary prevention plans have been published previously (63), and strategies that have been implemented at the state and local levels to address the problem also have been outlined previously (64). As ACCLPP noted, implementation of state and local primary prevention plans will require 1) targeting the highest risk areas, populations, and activities; 2) fostering political will for jurisdictions to provide an adequate level of funding; 3) expanding resources for housing remediation; identification and correction of lead hazards; and 4) establishing a regulatory infrastructure to create and maintain lead-safe housing and to support the use of lead-safe construction work practices (63,65). Links to state and local health department web sites, which include their primary prevention plans, are available at http://www.cdc.gov/nceh/lead/grants/contracts/CLPPP%20map.htm. Certain state and local health departments initiate case management services and home inspections when BLLs reach 10 mg/dL. As more primary prevention strategies are implemented, the number of health departments pursuing home inspections when BLLs reach 10 µg/dL will likely increase. Certain communities have developed online registries to help parents identify homes that are lead-safe or that have lead hazards (66). Steps to Identify and Safely Reduce Lead-Based Paint Hazards in Homes Lead-based paint hazards in homes are important sources of lead exposure. Preventive actions can be implemented to identify and address these hazards. Tenants can request a copy of all lead testing reports for housing sites from landlords at any time. Their landlord should have been provided with such information when they purchased the building; compliance with a tenant request for a copy of all lead testing reports is required by federal law (67). In addition, federal regulations require sellers and landlords 1) to disclose the possible presence of lead-based paint in any pre-1978 property and 2) to provide information on known lead-based paint and lead-based paint hazards at the time final agreements are signed on the purchase or rental of most housing built before 1978 (e.g., by providing results of any past evaluations of the property for lead) (67). Prospective buyers or renters have the opportunity to arrange for a lead inspection or risk assessment by a qualified professional at their own expense; buyers have up to 10 days to check for lead. Further, the law requires sellers, landlords, and renovators to provide buyers, renters, and those hiring renovators with an EPA-approved pamphlet, "Protect Your Family from Lead in Your Home" (68). To protect their children from lead, parents might choose not to buy or rent a property or to negotiate remediation of identified lead hazards. However, landlords or homeowners might not know whether their property has any lead-based paint or lead hazards. Lead-based paint hazards are likely to be present in older homes; all homes built before 1978 should be presumed either to have a lead hazard present or to contain intact lead-based paint unless a licensed lead inspector has determined otherwise. Lack of a deteriorated surface decreases the likelihood of lead-contaminated dust being present but does not ensure its absence. Knowledge of general characteristics of lead-based paint and lead-based paint hazards and their control might help parents to understand their home better (Box) (69--73). Screening for lead dust hazards through dust wipe testing (i.e., standardized collection of dust by wiping surfaces and measurement of lead collected) can help identify areas of concern. Because lead is not distributed uniformly within a home, wipe testing neither ensures absence of lead hazards at locations in the home that were not tested, nor does it ensure future protection from lead dust hazards if lead-painted surfaces subsequently deteriorate or are disturbed. Potential sources of future contamination include lead-containing paint on areas disturbed by impact/friction (e.g., windows, doors, and floors) and the interior migration of lead-contaminated exterior dust and soil (70). However, identifying lead dust hazards in the home is a first step toward protecting children and might help parents lower lead dust levels in their homes (74). Proper training is recommended for those collecting dust wipes to focus tests on areas at highest risk (63). Parents or property owners who wish to perform dust wipe sampling may consult their local health or housing departments for advice regarding sampling procedures, interpretation of results, and further actions based on results. For a lead-safe environment to be established in older buildings, repair of lead hazards and careful attention to maintenance is necessary. However, local ordinances typically do not require action until a child's BLL is elevated, and property owners might be unaware of lead hazards or ignore them. Primary prevention is possible only if the focus on safety in older housing is increased and lead hazards are repaired proactively before a child is exposed. In all pre-1978 properties, owners should use lead-safe work techniques when implementing routine maintenance to decrease the likelihood of lead hazards developing in a home. Home renovation or repair is known to be a risk factor for increasing or elevated BLLs, principally through exposures to the dust residue generated during the work (35,75--77). All contractors who perform repair and renovation work in older housing should be trained in lead-safe work practices and comply with any state and local requirements governing work with lead paint hazards (78). Property owners doing work themselves should seek expert advice and training to protect themselves and their families (79,80). Lead-safe work practices include 1) relocating families when the work warrants, 2) minimizing the amount of dust created, 3) containing dust in the work area, 4) cleaning up completely, 5) disposing of waste safely, and 6) performing clearance testing (i.e., testing of dust for lead after site clean up) to ensure that residual lead levels do not exceed EPA standards (81). Families with young children should be restricted from work areas until clearance testing has been performed and the area has been judged safe. In previous evaluation studies, lead dust clearance standards were not low enough to protect children from increased exposures to lead-contaminated dust after lead hazard remediation; as a result, after home repairs, BLLs of children with preremediation BLLs <25 µg/dL increased (82). In 2001, the EPA's lead dust clearance standards were lowered to 40 µg/ft2 for floors, 250 µg/ft2 for windowsills, and 400 µg/ft2 for window wells (81). No studies have evaluated if these lower clearance levels protect children whose BLLs are <10 µg/dL adequately from ongoing lead exposure. A cross-sectional study estimated that 20% of children with a current exposure to floor dust-lead at 40 µg/ft2 will have BLLs >10 µg/dL (83). A study conducted in 1994--1999 in 14 U.S. cities involving 2,682 pre-1978 homes demonstrated reductions in lead dust levels and fall in children's BLLs when lead-safe work practices were used during remediation efforts (69,84,85). The study applied lead dust clearance standards substantially less stringent than those currently in place, although clearance floor dust lead levels were generally low (geometric mean: 16 µg/ft2) (86). However, among the 869 children in this study who were tested within 4 months before home lead remediation and approximately 7 weeks after remediation, 81 (9.3%) had a clinically significant increase (>5 µg/dL) in BLLs; infants, children of less-educated mothers, and children from homes with higher numbers of preintervention exterior lead hazards were at highest risk (87). Dust lead levels at clearance were not associated significantly with an increase in BLLs. The study listed multiple types of exposures (e.g., other homes, parental job exposures) that might have accounted for increasing BLLs, but these were not evaluated systematically. Although lead remediation work reduced overall lead dust and BLLs, the finding that >9% of children had a rise in BLL of >5 µg/dL underscores the need to maintain a high level of vigilance to ensure that children are protected when homes or apartments undergo renovation and repair. Educational Strategies Lead exposure prevention strategies for children with BLLs <10 µg/dL typically focus on education and promotion of home cleanliness, without further identifying lead hazards or repairing them. Providing low-income parents with lead-related education via video in a pediatric office has been demonstrated to be effective in increasing knowledge and parental report of compliance with lead prevention actions in the home (88). No studies have evaluated office-based education with accompanying in-home strategies or used children's BLLs as the outcome measure for an office-based education strategy. Studies of children at high risk that applied intervention strategies in the home or community have demonstrated the failure of education and nonprofessional cleaning conducted alone (i.e., in the absence of other measures to reduce lead exposure) in preventing the development of BLLs >10 µg/dL (2,89--91). Few studies have applied prospective designs that included control groups. One study indicated that a highly intensive education program starting at birth and lasting for >3 years (28 sessions) delivered by community members lowered the risk of BLLs >10 µg/dL 34%, but this result was not statistically significant (92). Repeated in-home lead prevention education, even when accompanied by complimentary supplies of cleaning materials, was ineffective in lowering the incidence of elevated BLLs (93,94). A review of four studies (90) involving caregiver education (94,95) and professional house cleaning (96,97) indicated that such low-cost interventions reduced the overall proportion of children with BLLs >15 µg/dL or >20 µg/dL, but the effect on mean BLLs was not statistically significant (p>0.05). Intensive cleaning regimens reduce lead levels; in one study, biweekly professional cleaning resulted in a 17% decrease in mean BLLs after 1 year (96). However, the benefit of such intense and repeated cleaning was limited to homes without carpets (98). Intense cleaning can be used without subjecting children to a risk for increased lead exposure from unsafe repair methods (i.e., those not in compliance with lead-safe work practices). A single intensive cleaning alone does reduce levels of lead in dust by 32% to 93% depending on surface tested and starting lead concentration (99), but reaccumulation occurs within 3--6 months (100,101). A study that involved children with BLLs 15--19 µg/dL compared the effects of nurse home visits (five visits during 1 year) accompanied by lead dust tests with those of usual care (one or two visits by an outreach worker during 1 year) (74). After 1 year, dust lead levels were significantly lower (p<0.05) in homes where lead dust tests had been conducted during intervention than in usual care homes. This finding suggests that dust testing might help parents better understand lead hazards and take action to lower them. However, changes in dust lead were not mirrored by changes in BLLs in this group of children with elevated BLLs. Blood Lead Screening StrategiesCDC (102) and AAP (3) have recommended that health-care providers conduct blood lead tests on children enrolled in Medicaid and those identified as being at risk on the basis of the state or local screening plan or the risk assessment process. Federal policy requires that all children enrolled in Medicaid receive screening blood lead tests at ages 12 and 24 months and that blood lead screening be performed for children aged 36--72 months who have not been screened previously (103). Despite this, blood lead screening rates for Medicaid children have been low (<20%) (104) and in certain areas remain at approximately 20% (105). In 1997, CDC requested state and local health officials to use local communitywide data (e.g., BLL prevalence, housing age, and poverty status) to develop plans for blood lead screening for their jurisdictions and provide them to clinicians (102). These plans recommend either universal or targeted blood lead screening. State and local screening plans are available at http://www.cdc.gov/nceh/lead/grants/contacts/CLPPP%20Map.htm. Targeted screening strategies enable clinicians to assess risks for individual children and recommend blood lead testing for a subset of children in the jurisdiction thought to be at increased risk for lead exposure. CDC recommends that risk evaluations be conducted on the basis of such factors as residence in a geographic area, membership in a group at high risk, answers to a personal-risk assessment questionnaire (which might include local factors such as cultural practices or products, such as herbal remedies, traditional cosmetics or imported spices), or other risk factors relevant to the jurisdiction (102). CDC recommends that locally developed targeted risk assessment and blood lead screening strategies be applied at ages 1 and 2 years (102). Children aged 36--72 months who have been identified as being at risk and who have not been screened previously also should receive a blood lead test (102). For clinicians in areas that lack a state or local screening plan, CDC recommends that a blood lead test be performed on all children at ages 1 and 2 years and on children aged 36--72 months who have not been screened previously (102). Because lead exposures might change with a child's developmental progress (e.g., walking or reaching window sills) or as a result of external factors (e.g., family relocation or home remodeling), two routine screenings are recommended (at approximately ages 1 and 2 years). Among children in Chicago at high risk with BLLs <10 µg/dL at age 1 year, 21% had a BLL of >10 µg/dL when tested again at age >2 years (103). This report does not change current CDC recommendations in ages for routine blood lead testing. However, certain local health departments (e.g., those in Chicago, Illinois; New York, New York; and Philadelphia, Pennsylvania) recommend blood lead screening at younger ages or more frequently (106--108). For example, these departments recommend BLL testing starting at ages 6--9 months in high risk areas, blood lead testing at more frequent intervals (e.g., every 6 months) for children aged <2 years, or the provision of additional education and more rapid follow-up blood lead testing for children aged <12 months with BLLs 6--9 µg/dL. Personal Lead Risk Assessment QuestionnairesThe effectiveness of personal risk assessment questionnaires in identifying children with elevated BLLs has been documented in the scientific literature (Table 2) (109--125). However, no studies have evaluated the performance of these questionnaires at cut-off levels <10 µg/dL or their effectiveness in directing counseling or in identifying lead hazards in the home. When applied in consecutive samples of patients in clinical settings, the sensitivity of such questionnaires to identify children with BLLs >10 µg/dL varies considerably by population (109--128). In certain studies, the sensitivity improved if higher cut-off levels were used in the analysis (103,115,119,120) or if the questions used were developed specifically for the population tested (113,116,117,119,120,122). In general, to identify approximately 80% of children with BLLs >10 µg/dL, a blood test had to be performed for more than half of those children whose risk factors for lead exposure were assessed using a questionnaire. Multiple studies in populations with low (109,110,112--114, 127,128) or high (123,124) prevalence for elevated BLLs concluded that risk assessment questionnaires were not effective in their clinical settings. Future Research NeedsFurther study is needed to assess the effects of BLLs <10 µg/dL on children. Such research will entail following large and diverse populations, with careful attention to potential confounders and measurements of social factors. Additional research also is needed to evaluate the effectiveness of strategies to lower exposures to lead. This should include research on the effectiveness of strategies applied in the medical office and home and those that provide interventions through medical, public health, and environmental means. Blood lead screening strategies should be evaluated to determine the most appropriate ages for screening and the utility of screening strategies applied at the community level. Evaluations of lead surveillance strategies should test ways to identify changing patterns of environmental risks and subpopulations exposed to established and emerging sources of lead. In addition, better ways should be identified to alert public and clinical health-care professionals of changes in exposure sources and patterns and to enhance their response to such changes by increased surveillance and blood lead monitoring of populations identified as being at increased risk for exposure. Additional studies might provide data that can be used to improve laboratory methods and performance monitoring. This will require developing criteria to evaluate individual laboratories and mechanisms to provide this information to clinicians. Summary of RecommendationsFor Clinicians
For Government Agencies
AcknowledgmentHelpful suggestions were provided by Patrick J. Parsons, PhD, Lead Poisoning/Trace Elements Laboratory, Wadsworth Center, New York State Department of Health, Albany, New York. References
* A list of members of this committee appears on page 16 of this issue. † A complimentary video or DVD entitled, "CDC Guidelines for Collecting and Handling Blood Lead Samples---2004," may be obtained from the National Center for Environmental Health, Division of Laboratory Sciences, Lead and Multielement Proficiency Program at e-mail [email protected].
Advisory Committee on Childhood Lead Poisoning Prevention Membership List, October 2004--February 2006 Chairperson: Carla Campbell, MD, Children's Hospital of Philadelphia, Philadelphia, Pennsylvania. Executive Secretary: Mary Jean Brown, ScD, Division of Environmental and Emergency Health Services/Agency for Toxic Substances and Disease Registry, National Center for Environmental Health, CDC, Atlanta, Georgia. Members: Magaly C. Angeloni, MBA, Rhode Island Department of Public Health, Providence, Rhode Island; Valerie Charlton, MD, California Department of Health, Richmond, California; Walter S. Handy, Jr., PhD, Cincinnati Health Department, Cincinnati, Ohio; Ing Kang Ho, PhD, University of Mississippi Medical Center, Jackson, Mississippi; Valarie Johnson, Urban Parent to Parent, Rochester, New York; Linda Kite, MBA, Healthy Homes Collaborative, Los Angeles, California; Jessica Leighton, PhD, New York City Department of Health and Mental Hygiene, New York City, New York; Sally Odle, SafeHomes, Inc., Waterbury, Connecticut; George G. Rhoads, MD, University of Medicine and Dentistry of New Jersey, Piscataway, New Jersey; Catherine M. Slota-Varma, MD, Medical College of Wisconsin, Milwaukee, Wisconsin; Wayne R. Snodgrass, MD, PhD, University of Texas Medical Branch, Galveston, Texas; Kevin U. Stephens, Sr., MD, JD, New Orleans Department of Health, New Orleans, Louisiana; Helen J. Binns,* MD, Feinberg School of Medicine, Northwestern University, Chicago, Illinois; Kimberly M. Thompson,† ScD, Harvard University, Boston, Massachusetts. Nonvoting Federal Members: Phyllis Stubbs-Wynn, MD, Maternal and Child Health Bureau, Health Resources and Services Administration, Washington, DC; Michael Bolger, PhD, U.S. Food and Drug Administration; Washington, DC; John Borrazzo, PhD, U.S. Agency for International Development, Washington, DC; David Jacobs,§ PhD, U.S. Department of Housing and Urban Development, Washington, DC; Warren Friedman, PhD, U.S. Department of Housing and Urban Development, Washington, DC; Jacqueline E. Mosby, MPH, U.S. Environmental Protection Agency, Washington, DC; Walter Rogan, MD, National Institute of Environmental Health Sciences, Washington, DC; Robert J. Roscoe, MS, National Institute for Occupational Safety and Health, CDC, Cincinnati, Ohio; Lori E. Saltzman, MS, U.S. Consumer Product Safety Commission, Washington, DC; Jerry Zelinger, MD, Center for Medicare and Medicaid and Services, Washington, DC. Nonvoting Liaison Representatives: Steve M, Hays, American Industrial Hygiene Association, Nashville, Tennessee; Ezatollah Keyvan-Larijani, MD, DrPH, Council of State and Territorial Epidemiologists, Baltimore, Maryland; Pat McLaine,¶ MPH, National Center for Healthy Housing, Columbia, Maryland; Jonathan Wilson, MPP, National Center for Healthy Housing, Columbia, Maryland; Benjamin Gitterman, MD, American Public Health Association, Washington, DC; Routt Reigart II,** MD, American Academy of Pediatrics, Charleston, South Carolina; George C. Rodgers, Jr., MD, PhD, American Association of Poison Control Centers, Georgetown, Indiana; Jan Towers, PhD, American Academy of Nurse Practitioners, Gettysburg, Pennsylvania; Anne M Guthrie, MPH, Alliance for Healthy Homes, Washington, DC; Calvin B. Johnson, MD, American State and Territorial Health Officials, Harrisburg, Pennsylvania. * Member 2002--2004. . † Member 2002--2005. § Member 1996--2004. ¶ Representative 1998--2005. ** Representative 1997--2004. Table 1 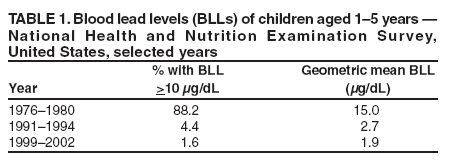 Return to top. Table 2 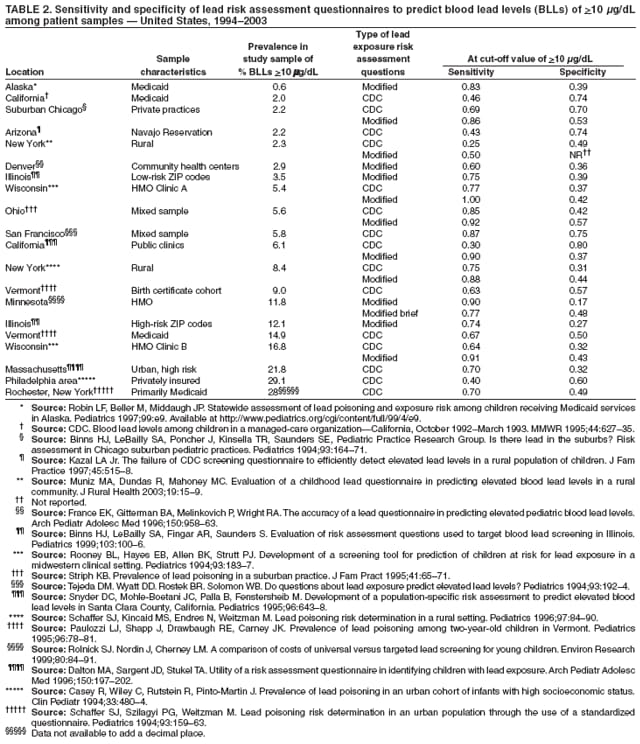 Return to top. Box  Return to top.
Disclaimer All MMWR HTML versions of articles are electronic conversions from ASCII text into HTML. This conversion may have resulted in character translation or format errors in the HTML version. Users should not rely on this HTML document, but are referred to the electronic PDF version and/or the original MMWR paper copy for the official text, figures, and tables. An original paper copy of this issue can be obtained from the Superintendent of Documents, U.S. Government Printing Office (GPO), Washington, DC 20402-9371; telephone: (202) 512-1800. Contact GPO for current prices. **Questions or messages regarding errors in formatting should be addressed to [email protected].Date last reviewed: 10/10/2007 |
|||||||||
|